What Does the IRB Review?, Research
Por um escritor misterioso
Descrição
Below are the elements the IRB looks for when reviewing research. Federal regulations 45 CFR 46.111 and 21 CFR 56.111 outline the requirements for approval of non-exempt human subjects research. To obtain IRB approval, the IRB must have enough information to determine the criteria in each of the sections below are satisfied.

IRB Review Timeline - Office of Research Support and Compliance

Requirements for Institutional Review Board (IRB) Review and HIPAA Waiver Documentation for RIF DUA Request Submissions
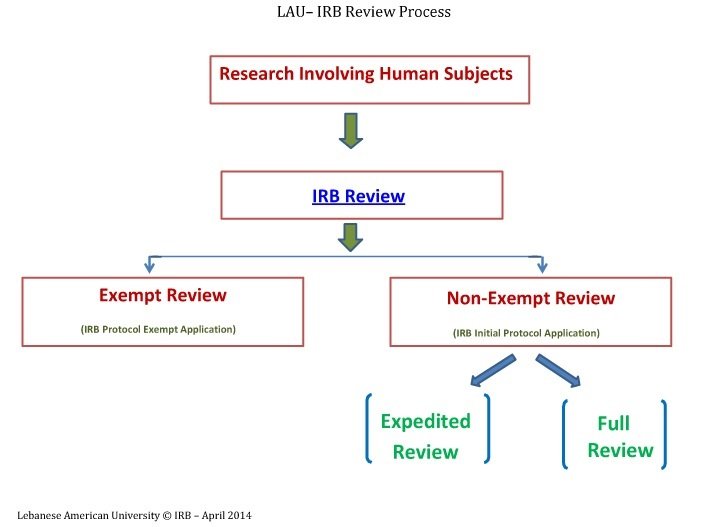
IRB Types of Review, Ethical Compliance, Graduate Studies and Research

PDF] The purpose, composition, and function of an institutional review board: balancing priorities.
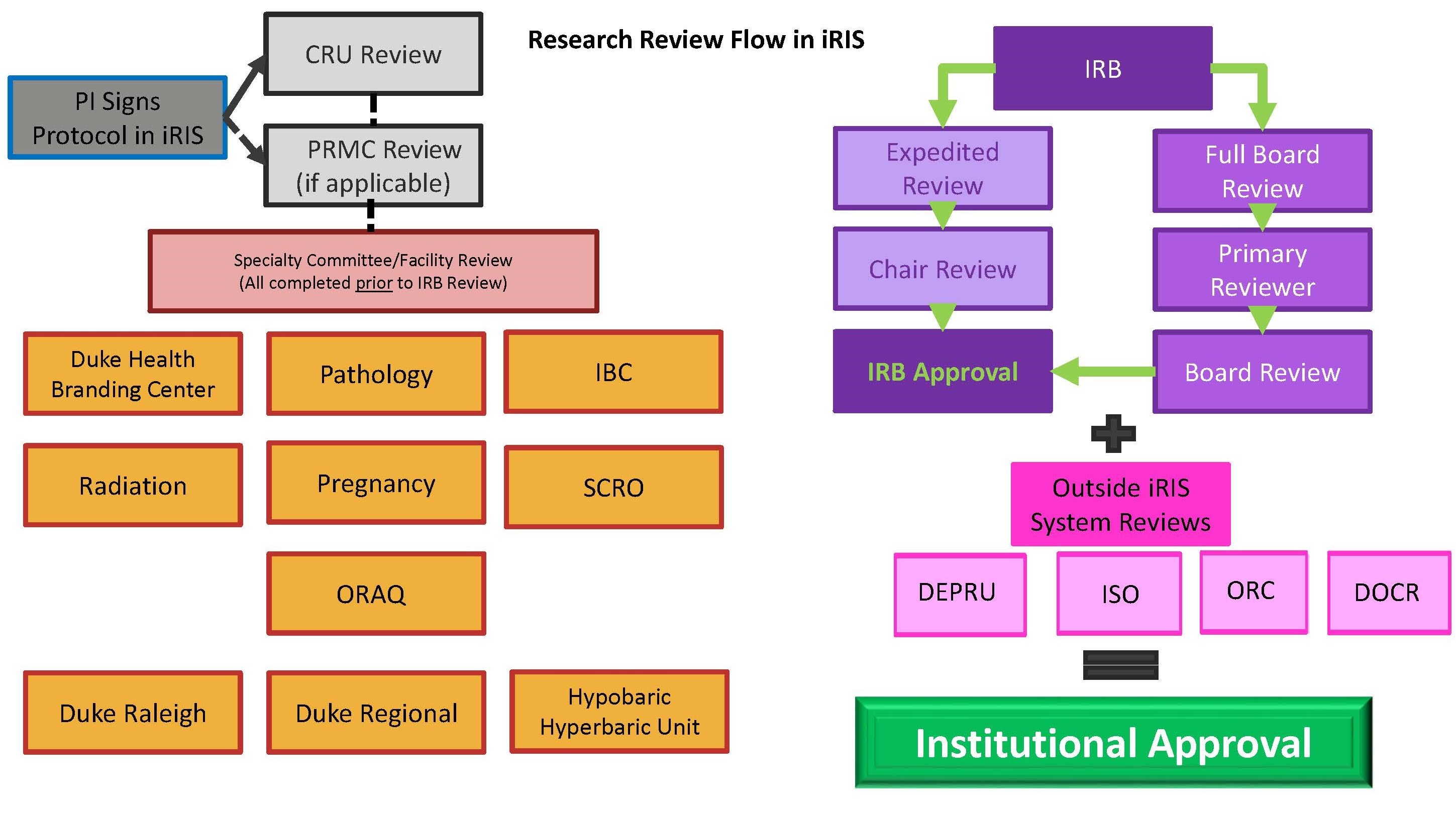
IRB Process Duke Health Institutional Review Board
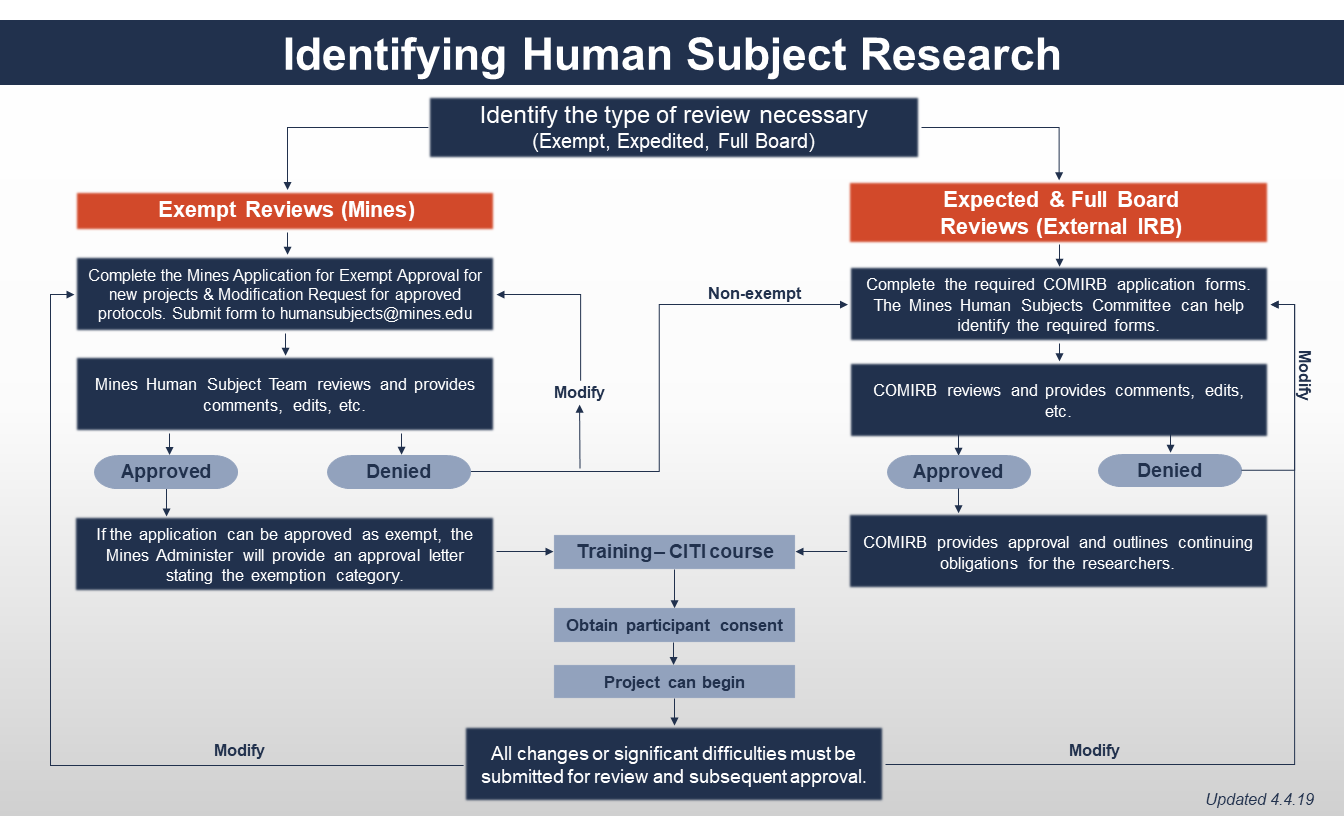
Policy & Procedures - Human Subjects Research

Levels of IRB Review
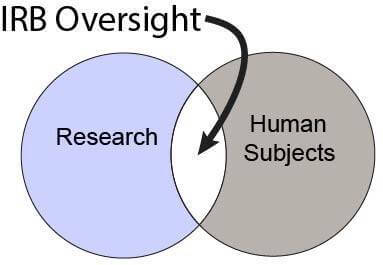
Is IRB Approval Required?

IRB reliance: An informatics approach - ScienceDirect

What is the Institutional Review Board? — Delve

Ethics in Research: What is the IRB? – ScIU
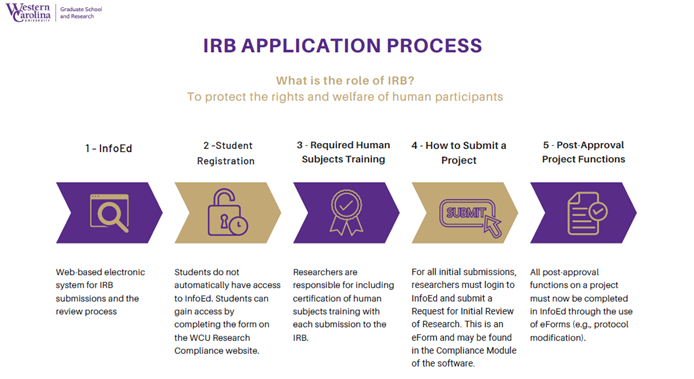
Western Carolina University - Institutional Review Board FAQs
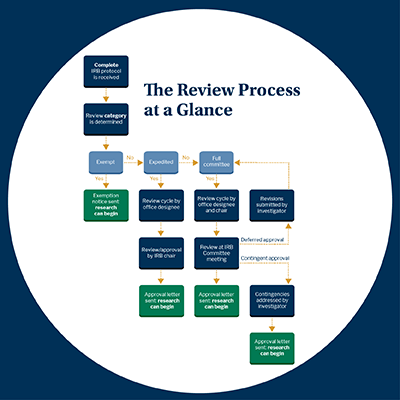
Institutional Review Board (IRB)
Doctoral Dissertation Research and the IRB, 2021, IRB Blog, Institutional Review Board
de
por adulto (o preço varia de acordo com o tamanho do grupo)







